Introduction
Perianal fistula-in-ano is a common anorectal disorder arising predominantly from cryptoglandular infection and represents a chronic manifestation of ongoing perianal sepsis originating from the infected anal glands. The extent and behavior of the fistulous tract are determined by its anatomical relationship with the anal sphincter complex, which underpins the distinction between simple and complex fistulas and has important implications for continence-preserving management.1,2 While the majority of fistulas remain confined to the perianal region, complex fistulas may extend along established fascial planes and involve adjacent anatomical compartments.2 Scrotal extension is uncommon and may dominate the clinical presentation, mimicking primary scrotal pathology and resulting in diagnostic delays and inappropriate initial management.3
Preoperative magnetic resonance imaging (MRI) is widely regarded as the reference standard for delineating the fistulous anatomy in complex diseases.4 However, in many low-resource settings, access to advanced imaging remains limited, and clinical decision-making may rely heavily on physical examination and intraoperative findings.5
Case Presentation
A 47-year-old man presented with progressive scrotal swelling associated with perineal pain, purulent discharge, intermittent fever before admission, nausea, and constipation. The patient reported a history of intermittent anorectal symptoms for approximately two years, during which he primarily sought alternative treatment, before experiencing progressive worsening over the two weeks preceding admission. There was no history of diabetes mellitus, tuberculosis, inflammatory bowel disease, or anorectal surgery.
On admission, he was hemodynamically stable but tachycardic, with a blood pressure of 116/70 mmHg, heart rate of 114 beats per minute, respiratory rate of 22 breaths per minute, body temperature of 36.6°C, oxygen saturation of 96% on room air, and a Glasgow Coma Scale score of 15. Laboratory evaluation revealed marked leukocytosis (21.6 ×10³/mm³; reference range, 4–11 ×10³/mm³) with neutrophil predominance (90.0%; reference range, 51–67%), mild anemia (hemoglobin 11.7 g/dL; reference range, 14–18 g/dL), and elevated urea level (66 mg/dL; reference range, 17–43 mg/dL). Other renal and liver function parameters were within the normal limits.
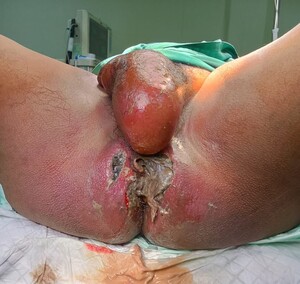
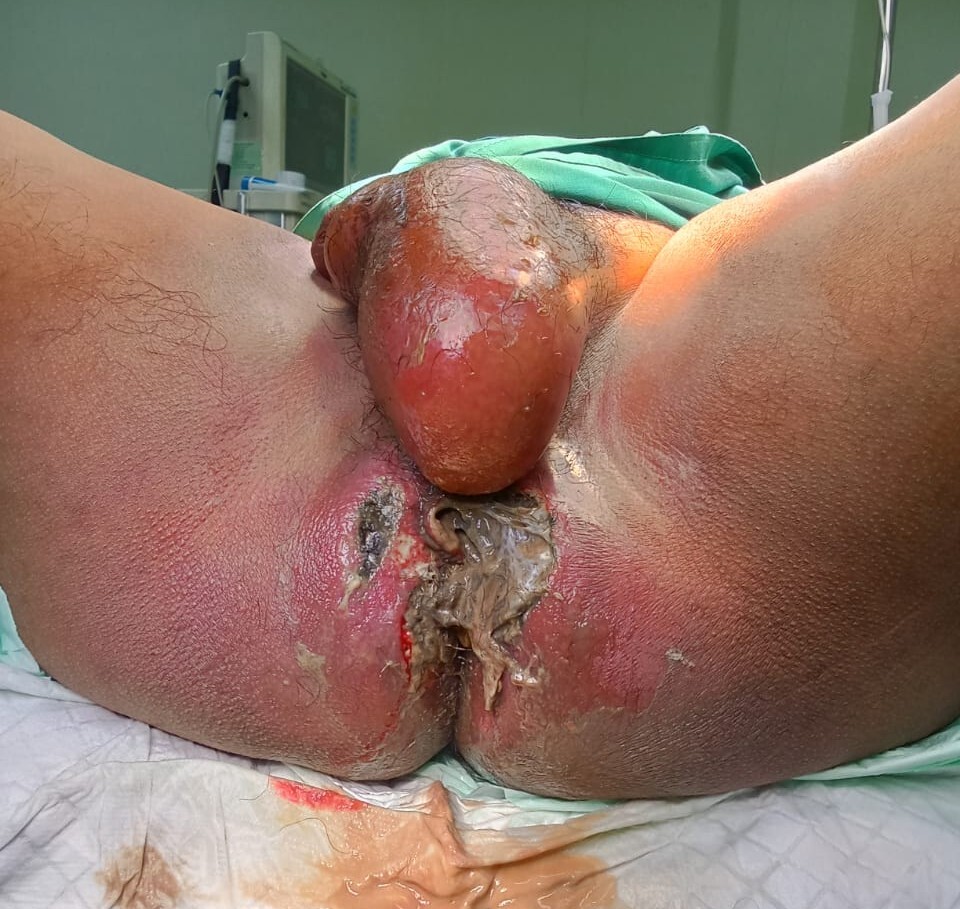
Local examination revealed extensive perineal tissue destruction with necrosis, erythema, fluctuations, and foul-smelling purulent discharge (Figure 1). Multiple coalescent external fistulous openings were present, precluding the clear identification of individual tracts. Scrotal examination showed marked edema and fluctuation consistent with abscess formation without evidence of inguinal hernia.
Advanced imaging studies were not performed because of limited availability; therefore, clinical decision-making relied on physical examination, and operative exploration was performed as the primary means of anatomical assessment.
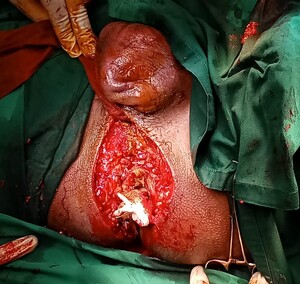
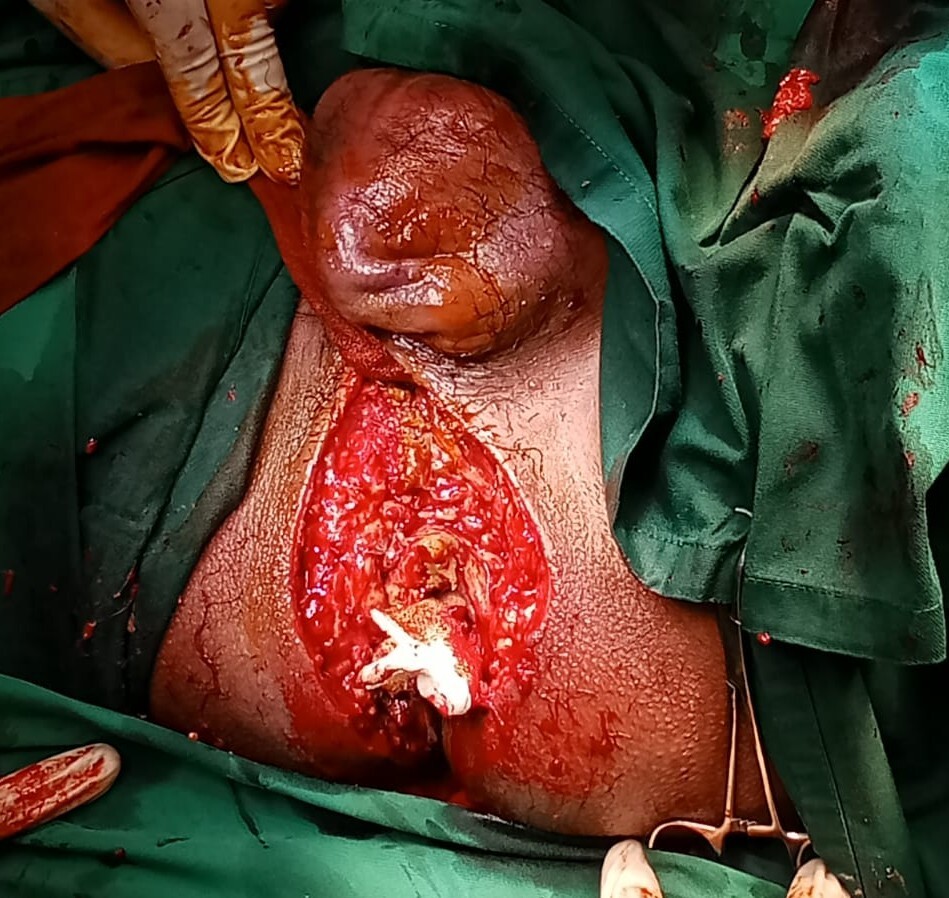
Emergency surgical exploration revealed a fistulous tract traversing the external anal sphincter, with anterior extension along the perineal fascial planes into the scrotum, consistent with a complex transsphincteric perianal fistula. Extensive necrotic tissue, multiple coalescent external openings, and a large scrotal abscess communicating directly with the fistulous tract were also identified. Sphincter involvement was assessed intraoperatively, and excision was limited to the infected and non-viable tissues. Complete fistulectomy, wide debridement of necrotic tissue, and drainage of the scrotal abscess were performed.
Histopathological examination of the perianal biopsy specimens revealed ulcerated skin with granulation tissue and extensive acute suppurative inflammation characterized by dense neutrophilic infiltrates and areas of necrosis. No granulomatous inflammation, dysplasia, or malignant cells were identified, consistent with cryptoglandular perianal fistula.
Postoperatively, the patient received daily wound care, and postoperative antibiotics were administered for six days, selected in accordance with local antimicrobial protocols. The postoperative course was uneventful and there were no infectious or functional complications. The patient was discharged after six days of hospitalization and continued outpatient follow-up for more than one month, during which progressive wound healing and clinical improvement were observed.
Discussion
Scrotal extension of perianal fistulas is an uncommon manifestation of fistula-in-ano and has been reported only in a limited number of adult cases.3 In such presentations, genital symptoms may dominate the clinical picture, leading to initial misattribution as primary scrotal pathology and subsequent diagnostic delay. Similar to previously reported cases, the initial presentation in our patient was dominated by scrotal swelling and abscess formation, which obscured the underlying anorectal source of the infection.3,6 Recognition of this diagnostic pitfall is important because delayed identification of fistulous disease may permit ongoing sepsis and result in more extensive local tissue destruction.
The anatomical basis for the anterior extension of the perianal fistula can be explained by the continuity of the perineal fascial planes. Infection originating from the anal glands may track along the intersphincteric or transsphincteric pathways and spread anteriorly through the Colles’ fascia into the Dartos fascia of the scrotum, facilitating the formation of a communicating scrotal abscess.7,8 In the present case, the intraoperative findings were most consistent with a complex transsphincteric fistula, a classification that is clinically relevant given its implications for operative planning, risk of recurrence, and continence outcomes.1,9
MRI is widely regarded as the reference standard for delineating fistula anatomy, identifying secondary tracts, and guiding operative planning in complex fistula-in-ano.4,10 However, access to advanced imaging may be limited in certain settings. In such circumstances, careful clinical assessment combined with meticulous intraoperative exploration is essential. In the present case, surgical exploration served both diagnostic and therapeutic purposes, allowing the identification of sphincter involvement and fistulous extension while simultaneously achieving definitive source control.5,11
Management of complex fistula-in-ano remains heterogeneous, reflecting the absence of a universally accepted optimal approach.12 A wide range of operative strategies has been described, including staged drainage with seton placement, sphincter-preserving techniques, and definitive excisional procedures. While sphincter-preserving approaches are often advocated to minimize functional impairment, long-term outcome studies suggest that fistula complexity itself is a major determinant of recurrence and that sphincter-sparing procedures may be associated with higher recurrence rates without consistent long-term advantages in patient-reported quality of life.13 Consequently, operative decision-making should balance the risk of functional compromise with the need for effective eradication of sepsis.
In the present case, extensive local necrosis, multiple coalescent external openings, and a large scrotal abscess communicating directly with the fistulous tract rendered prolonged staged management impractical. Following individualized risk–benefit assessment, complete fistulectomy with wide debridement was performed to achieve definitive source control. Although the potential risk of continence was acknowledged, prioritization of infection control was considered necessary to prevent ongoing sepsis and further tissue destruction, an approach supported in complex fistula cases presenting with advanced local infection.13,14
The differential diagnosis of scrotal swelling accompanied by systemic inflammatory features is broad and includes Fournier’s gangrene, epididymoorchitis, hidradenitis suppurativa, and primary scrotal abscess. Fournier’s gangrene is of particular concern because of its aggressive nature and high mortality if not promptly treated. It is typically characterized by rapid progression, extensive fascial necrosis, severe systemic toxicity, and the need for repeated surgical debridement, features that were not observed in this patient.14 The absence of testicular tenderness or urinary symptoms, together with intraoperative demonstration of direct anorectal communication, further supports the exclusion of alternative diagnoses.6
This case highlights the influence of delayed healthcare-seeking behavior and limited diagnostic resources on disease severity at presentation. In environments with constrained resources, surgical practice often necessitates adaptation to infrastructural limitations, with greater reliance on clinical judgment and intraoperative findings rather than idealized diagnostic pathways. Such contextualized decision making reflects real-world surgical practice in many settings and remains essential for delivering effective care when advanced imaging and comprehensive diagnostic support are unavailable.5,11 Therefore, early consideration of an anorectal source in patients presenting with atypical scrotal infections is critical to prevent diagnostic delay and associated morbidity.
Conclusion
Perianal fistulas rarely present as scrotal abscesses, mimicking primary scrotal pathology. In the absence of advanced imaging, intraoperative findings play a critical role in establishing diagnosis and guiding management. Early consideration of the anorectal source and timely surgical exploration are essential to prevent ongoing infection and morbidity, particularly in resource-limited settings.
Ethics approval
Written informed consent was obtained from the patient for the publication of this case report.
Availability of data and material
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors
Authors’ contributions
II conceived the study and drafted the manuscript. MRDS contributed to drafting and manuscript review. AHR supervised the study and was responsible for validation and resources, and all authors contributed substantially to its revision.
Acknowledgements
None