Introduction
Asthma is a chronic respiratory condition characterized by the following features: airway hyperresponsiveness, airway thickening, reversible bronchoconstriction, increased mucus production and variable expiratory airflow limitation. Patients experiencing asthma exacerbations typically experience the following symptoms: shortness of breath, wheezing, chest tightness and cough. The management of asthma has been shown to be highly responsive to inhaled corticosteroids because of the underlying inflammatory pathophysiology of asthma, which will be further discussed below. In fact, inhaled corticosteroids have been the backbone of asthma therapy for decades. The dosing ranges for each of these agents are described by the Global Initiative for Asthma and are shown in Figures 1 and 2.1 The benefits of their use have been widely studied and have been repeatedly shown to be beneficial for asthmatic patients by reducing the frequency and severity of their symptoms, improving overall lung function and reducing the frequency of asthma related hospitalizations.2 However, little research has been conducted regarding the possible adverse effects of long-term exposure to high-dose inhaled corticosteroids, despite existing evidence showing that many patients must escalate their dosages owing to inadequate improvement in their symptoms.3 Even fewer investigations have been conducted to analyze whether prolonged exposure to high-dose ICSs is beneficial for improving lung function to justify the possible adverse events that can occur. The purpose of this paper is to analyze existing evidence to answer these two prevalent questions regarding the management of this unique patient population.
Pathophysiology of asthma
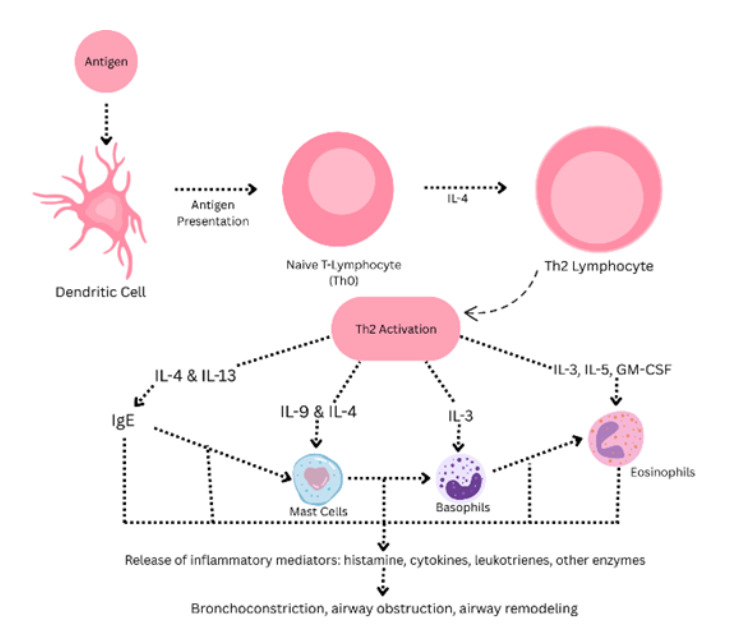
As discussed previously, asthma is best described as a chronic and inflammatory condition characterized by inflammation of the airway, lower airway obstruction, vari- able expiratory airflow limitation, airway hyperresponsiveness, airway thickening and increased sputum production.4 A characteristic feature of asthma is bronchocon- striction. This response is elicited by either methacholine or histamine on the basis of various triggers. Common asthma triggers include the following: cold air, cigarette smoke, exercise, allergen exposure, and viral upper respiratory tract infections.4 At the physiological level, there is a widely agreed upon mechanism that leads to the devel- opment of airflow obstruction. This mechanism is largely dependent on IgE-mediated inflammation. The entire inflammatory cascade in asthma is dependent upon a partic- ular terminally differentiated helper T-cell in the lungs of asthmatic patients, T helper type 2 (TH2) cells. The TH2 cells are responsible for the secretion of interleukins 4, 5, 9 and 13. All these interleukins contribute to the development of characteristic asthmatic changes.5 The first TH2 cell mechanism leads to an immediate hypersen- sitivity/type 1 hypersensitivity reaction. This reaction is mediated by immunoglobulin E (IgE). IgE is released by B-cells that are activated by IL-4 produced from TH2 cells. Circulating IgE is subsequently picked up by immunoglobulin receptors (Fc e recep- tor I [FceRI]). These receptors are located on numerous immune effector cells such as basophils, mast cells, eosinophils and other lung cells. After initial antigen exposure, subsequent antigen presentation causes FceRI receptor cross-linking. This will then activate immune effector cells and will cause them to undergo degranulation, releasing factors such as histamine which promote bronchoconstriction.5 A summary of the TH2 cell mediated pathway is presented below in Figure 3.
Mechanism of action of inhaled corticosteroids in asthma
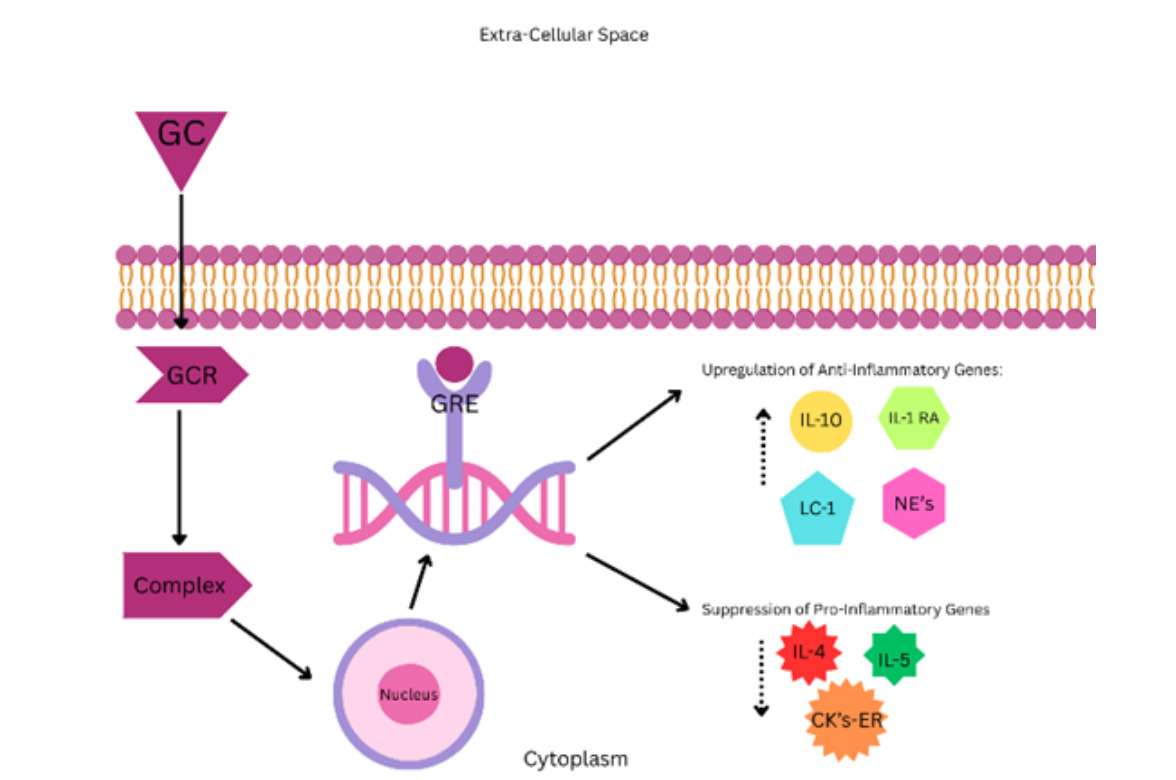
This will be a short summary to familiarize the reader of the mechanisms of ICS in relation to asthma to better conceptualize the topic at hand. However, this is not an exhaustive description, as it would be outside the scope of this paper. Inhaled corticosteroids are beneficial in asthma, because they interfere with the mechanisms by which an asthma episode develops. The mechanism of action of glucocorticoids starts with the binding of the glucocorticoid to the glucocorticoid receptor in the cell’s cytoplasm. Next, the receptor-substrate complex moves to the nucleus where it binds to DNA at the glucocorticoid response element. Thus, glucocorticoids are able to exert their anti-inflammatory effects by increasing the transcription and expression of anti-inflammatory proteins such as the IL-1 receptor antagonist, lipocortin-1, IL-10 and neutral endopeptidases. Most importantly, however, the anti-inflammatory effect can be attributed to the suppression of proinflammatory genes discussed previously (IL-4, 5 and chemokines involved in eosinophil recruitment), which are integral to the pathophysiology of asthma.5 Furthermore, ICSs can block the activation and recruitment of inflammatory cells such as eosinophils, dendritic cells, T lymphocytes and macrophages. While ICSs do not directly inhibit the activation of mast cells, they interfere with their ability to survive at the airway surface, which indirectly interferes with their ability to be activated.5 In summary, the molecular effects of ICSs are able to limit the progression of asthma by directly interfering with the inflammatory nature of this condition. This limits the degree of pathological changes that occur with asthma, thereby reducing the symptom burden, and frequency of exacerbations and improving lung function.
Adverse effects of high dose inhaled corticosteroids
The potential adverse effects of high dosages of ICSs are numerous and they have been investigated thoroughly. The mechanisms by which they occur will be discussed in a later section, but the etiology of these adverse effects is multifactorial. The nature of these adverse effects are strikingly similar to the adverse effects associated with systemic glucocorticoids.
Cardiopulmonary risks
A recent investigation reported an increased incidence of adverse cardiovascular and pulmonary events that occurred while patients were on a moderate to high dose ICS. These adverse cardiopulmonary events included the following: pneumonia, pulmonary embolism, arrhythmias, and cardiovascular events. These adverse events were not observed in patients on a low dose ICS. The authors further suggest that clinicians utilize the lowest effective possible dose of ICS to minimize potential risks.7
Endocrine Risks
In another study it was reported that 7.7 percent of patients on high-dose ICSs pre- sented signs of HPA-axis suppression. The authors supported the practice of adding a second agent such as a long-acting-beta-agonist (LABA) to minimize the dose of ICS. The danger of adrenal insufficiency in patients receiving systemic corticosteroids is a widely recognized issue and is at the forefront of most clinicians’ minds when initiat- ing these agents. However, due to the high systemic bioavailability of high dose ICSs, especially traditional agents, it is reasonable to conclude that HPA axis suppression occurs through a similar mechanism in ICSs as it does in systemic corticosteroids.8
Ophthalmologic risks
A meta-analysis was conducted to analyze the risk of cataract development in patients receiving high-dose ICS. There was a statistically significant association between high- dose ICS use and cataract formation. Like other adverse effects related to high-dose ICS, this is an issue that has been noted in patients who are chronically on systemic steroids. This suggests that there is a greater degree of systemic bioavailability in high dose ICS than was previously thought.9
Growth risks
There is also a statistically significant risk for growth retardation in children who were placed on high-dose ICS.10 The risk of growth retardation was reduced by using the lowest dose of inhaled corticosteroid possible. The risk was further reduced by making the ICS less systemically bioavailable via the use of an appropriate inhalation device.10 This is an issue that is at the forefront of most parents’ minds when considering therapy. Parents’ concerns about this development could contribute to therapy noncompliance and worsening of the patient’s condition. This further emphasizes the importance of risk mitigation with these agents.
Metabolic risks
Regarding the effects that high dose ICSs can have on metabolism, a study reported a 34 percent increased risk of developing diabetes in patients placed on ICSs, particularly patients who are taking high-dose ICSs.11 This is a known and well-studied complication associated with systemic glucocorticoids; however, owing to the moderate systemic bioavailability seen with high-dose ICSs, clinicians should also be cautious of this potentially adverse effect. This is particularly true in patients who are metabolically compromised at baseline.
Risks to bone mineral density
It was also demonstrated that both pre and postmenopausal women were at greater risk for the development of osteoporosis when exposed to high dose beclomethasone (750-1500 ug/day) for at least three months.12 As with other adverse events related to high dose ICS use, the mechanisms by which this occurs are identical to those of systemic glucocorticoids. Clinicians should be cautious when increasing the dose of ICS in those who are already predisposed to or have a history of osteopenia or osteoporosis to further reduce the risk of fracture.
Mechanism of adverse effects related to ICS use
Systemic bioavailability of traditional ICS agents
It has long been known that more traditional ICS agents, particularly fluticasone propionate, have a greater degree of systemic bioavailability than newer agents such as ciclesonide. This is likely why there are greater systemic adverse events associated with fluticasone propionate than with ciclesonide. The systemic bioavailability of ICSs depends on a few factors. The first factor to consider is the clearance rate of the ICS agent. Compared with more traditional agents, des-ciclesonide has a clearance rate of 228 L/hour.13 This rate comparable to the fluticasone propionate clearance rate of 65.58 L/hour or 1093 mL/min.14 The clearance rate of a drug is inversely proportional to the degree of its systemic bioavailability. Another factor to consider will is volume of distribution (Vd) of the ICS agent. In short, a drug with a larger Vd will have a greater distribution of the drug outside of the central compartment. An FDA biochemical review of ciclesonide revealed that it had a Vd of 2.9 L/kg.15 This value contrasts with fluticasone propionate which has a Vd of 4.2 L/kg.14 These findings were replicated in an experimental study that revealed that fluticasone propionate had a Vd of 9800L, whereas ciclesonide had a Vd of 947L.16 In summary, the systemic bioavailability of more traditional ICS agents, such as fluticasone, is greater than that of more novel agents like ciclesonide. It is highly probable that fluticasone propionate has a greater likelihood of adverse events than ciclesonide because fluticasone propionate has much greater systemic bioavailability.
Mechanisms of cardiopulmonary adverse events
The mechanism by which adverse cardiopulmonary events occur is not well understood, but there is a fair amount of experimental evidence that can provide clues. Systemic corticosteroids activate glucocorticoid receptors throughout the body and excessive activation can lead to the development of impaired glycemic control, hypertension and dyslipidemia.7 The development of these metabolic disturbances predisposes patients to the development of adverse cardiovascular events. However, a small amount of experimental research suggests that excessive activation of glucocorticoid receptors (which can be caused by high dose ICS exposure) can directly contribute to endothelial dysfunction, further predisposing people to adverse cardiovascular events.7 The risk of major adverse cardiovascular events was significantly increased in those with underlying cardiovascular disease who received high dose ICS. With respect to the development of cardiac arrhythmias, a study reported that patients receiving ICS were more likely to develop an arrhythmia than those who did not.17 The mechanism for this is also unclear. However, some evidence from animal model studies, (although not related to ICS directly but rather systemic methylprednisolone), has shown that large doses of corticosteroids can affect myocardial contractility and increase peripheral vascular resistance which can have a proarrhythmogenic effect.18 In patients receiving high dose ICS, there is a greater risk for the development of pulmonary embolisms. An experimental study revealed that asthmatic patients who were receiving a high dose of ICS had a procoagulant environment. In the same study even moderate ICS dosages were associated with the suppression of fibrinolytic factors, further predisposing these patients to experiencing a thromboembolic event.19 Finally, as far as pneumonia is concerned, there are a few suggested mechanisms for why high dose ICSs (particularly fluticasone) predispose people to pneumonia. These suspected mechanisms are based on previous research performed on COPD patients treated with systemic glucocorticoids but given the systemic bioavailability of more traditional ICS agents, these mechanisms can be easily translated to high dose ICSs. These mechanisms include the following: “modulation of the innate and adaptive immune system, increase in the bacterial load and changing the microbial composition of the airway”.7
Mechanisms of HPA-axis suppression
The mechanism by which the HPA axis is suppressed in ICS use is the same as that in systemic steroid use. Specifically, the administration of exogenous steroids suppresses the secretion of CRH from the hypothalamus via negative feedback. Furthermore, exogenous steroids interfere with the ability of CRH to stimulate the secretion of ACTH from the anterior pituitary. This eventually causes atrophy of the corticotrophin releasing cells in the anterior pituitary gland. As a result, the adrenal cortex loses the ability to produce cortisol, leading to HPA-axis suppression and adrenal insufficiency.20
Mechanisms of cataract development
The exact mechanism of cataract development is unknown. However, a substantial amount of research has been dedicated to discovering the etiology of steroid induced cataracts. One major influencing factor that can predispose patients to the development of steroid induced cataracts is intraocular growth factors. The specific growth factors that influence this process are as follows: FGF-2, insulin, insulin-like growth factor-1 (IGF-1), epidermal growth factor (EGF), transforming growth factor-beta (TGF-B), lens epithelium–de-rived growth factor (LEDGF), platelet-derived growth factor (PDGF), and bone morphogenetic proteins (BMPs). There is a particular point of interest in the role of FGF-2 and IGF-1. Glucocorticoids affect the concentration of FGF-2 in different ways based on the cell type being studied. In the cells of the anterior chamber, glucocorticoids decrease the concentration of FGF-2, which promotes lens epithelial cell proliferation. However, in the cells of the posterior chamber, glucocorticoids increase the concentration of FGF-2. This increased concentration will induce the posterior chamber cells to differentiate into fiber cells. The levels of IGF-1 are inversely proportional to the levels of FGF-2. Therefore, if there is an expected decrease in FGF-2, there will be an expected increase in IGF-1. IGF-1 is also related to lens epithelial cell proliferation and differentiation. Furthermore, mice that overexpress IGF-1 develop an “expanded equatorial transitional zone and perturbed lens polarization”.21 There are also other proposed mechanisms that lead to the development of cataracts in patients exposed to high dose steroids for a prolonged period. Some of these changes include the following: glucocorticoid induced modification of signal transduction, oxidation, lens hydration and protein steroid binding.
Mechanisms of growth inhibition and decreased bone mineral density
Like other adverse events related to high dose ICS, there is no difference between how high dose ICS and systemic corticosteroids/glucocorticoids exert these effects. Like systemic corticosteroids, high dose ICSs are potent inhibitors of linear growth. The mechanisms by which this occurs are numerous. These mechanisms include the following: “Blunting of pulsatile growth hormone release, down regulation of growth hormone receptor expression, inhibition of insulin-like growth factor-1 bioactivity and osteoblast activity, and suppression of collagen synthesis and adrenal androgen production”.22 All these factors decrease the linear growth velocity.
Mechanisms of metabolic and glycemic abnormalities
As discussed above, the mechanisms by which ICSs contribute to derangements in metabolism are the same as those of systemic corticosteroids/glucocorticoids. Like their systemic counterparts, ICSs will contribute to the development of impaired glycemic control and other metabolic abnormalities that increase a patient’s risk for developing diabetes mellitus. The way ICSs can do this is due to their systemic bioavailability. Specifically, with reference to the development of diabetes mellitus, there is an important focus on how glucocorticoids affect the function of pancreatic beta-cells. Glucocorticoids directly contribute to the development of pancreatic beta-cell dysfunction which results in a reduction in insulin secretion. The stimulation of glucocorticoid receptors in beta-cells leads to abnormal glucose metabolism. Specifically, it results in increased glucose cycling and reduced glucose transport secondary to the increased action of glucose-6-phosphatase. This eventually contributes to the development of pancreatic beta-cell dysfunction.23
Efficacy of high-dose vs low dose ICSs on exacerbation frequency and lung function
Low dose ICS remains the first-choice pharmacologic agent for asthmatic patients. There is significant evidence that low dose ICS not only reduces the number of emergency department visits24 but also significantly improves lung function. When low to medium dose ICSs are compared to high dose ICSs, there is no evidence of a significant improvement in the frequency of exacerbations or in lung function. There was nearly an identical improvement in lung function and the frequency of exacerbations when comparing the two dosages. Therefore, it cannot be recommended that a high dose of ICS be used when there is a risk for significant adverse events to develop, when clinical improvement can be seen when lower dosages are used.
Efficacy of high-dose ICS on exacerbation frequency
Numerous studies have suggested that low to medium dose ICSs are equally efficacious in reducing the frequency of exacerbations compared with high dose ICSs. Therefore, it is reasonable to conclude that lower doses of ICS can be used to control a patient’s asthma without the concern of developing adverse effects associated with higher dosages. Another study reported that high potency inhalers (medium dose ICSs or ICS-LABAs) were not superior to low dose ICSs in preventing exacerbations. Furthermore, a significant increase in the ICS dosage is unlikely to reduce exacerbations. In addition, there was a significant increase in FEV1 when low dose ICS was used.25 Zhang and colleagues reported that while patients are experiencing an asthma exacerbation, the use of increased versus stable ICS doses reduces the odds of requiring systemic corticosteroids for the exacerbation. However, there was no evidence for an impact on hospital admissions or unscheduled physician visits, and an increased ICS dose increased the risk of non-serious adverse events. This suggests that the marginal benefit of increasing the dosage of inhaled corticosteroids may not be worth the possible adverse effects associated with higher dosages.26 Another study revealed that low dose ICS-formoterol combination increased the probability of well controlled asthma compared with a substantial increase in the dose of an ICS.27 Like other studies, this study revealed the addition of another agent to the low dose ICS will adequately control a patient’s asthma without having to expose a patient to a higher-dose ICS. Similarly, Perng and colleagues reported that a low dose of an ICS plus a leukotriene receptor antagonist (LTRA) was as effective as a high dose of an ICS.28 Masoli M and colleagues showed that the addition of a LABA (salmeterol) to a moderate dose ICS resulted in significantly greater clinical benefit than increasing the dose of the ICS by twofold or greater.29 Finally, another study reported that escalation to high dose ICS was not effective at preventing future asthma exacerbations.30 In summary, escalation of the ICS dosage did not increase the probability of more well controlled asthma than low-dose ICS plus an additional agent.
Efficacy of high-dose ICS on lung function
With respect to lung function, Powell and colleagues reported that high-dose ICS was not associated with any significant improvement in lung function or asthma episode reduction compared with moderate-dose ICS. Compared to low dose ICS, moderate dose ICS was more effective as initial therapy. However, no benefit was noted when the ICS dose was doubled or quadrupled.31 In another study, there was a near maximal FEV1 increase in patients exposed to a low dose of fluticasone propionate (88 µg/day) and a medium dose of beclomethasone (672 µg/day). Furthermore, the near maximal improvement in bronchial hyperresponsiveness measured during the methacholine challenge occurred in low dose fluticasone propionate (88 µg/day) and beclomethasone (672 µg/day). In the high dose ICS arm, however, there was no significant improvementin lung function but there was greater overnight cortisol secretion.32
Rationale for maximal benefit at low–moderate doses
The maximum anti-inflammatory benefit occurs at low-moderate doses of ICS due to the relatively flat dose-response curve associated with these agents. At a mere 200-250 micrograms of fluticasone propionate (or equivalent), 80-90% of the maximum therapeutic benefit of this agent will be achieved.33 This is because of the underlying pharmacology associated with the glucocorticoid receptor occupancy. Low doses of inhaled corticosteroids can generate high glucocorticoid receptor occupancy rates of greater than 90% across the entire dose interval at steady state.34 Therefore, once receptor occupancy reaches a high level, there is little-no potential for improvement when the dosage is escalated.
Discussion
An evaluation of the current medical literature revealed that there is no significant clinical benefit when the dosage of inhaled corticosteroids is increased beyond a moderate dose. In addition, the limited benefits that are associated with high-dose ICSs are overshadowed by the significant adverse effects associated with high dose ICSs. The etiology behind the systemic adverse effects of high dose inhaled corticosteroids is likely rooted in systemic absorption. There is likely greater systemic absorption of high dose ICSs than previously believed, but future research should be conducted. Currently, whether more novel ICS therapies, such as Ciclesonide, show fewer systemic adverse effects than more traditional agents by acting as prodrugs that are activated by the respiratory epithelium is being investigated. This agent shows promising results, and further research should be undertaken to determine its efficacy when compared to traditional agents like fluticasone and beclomethasone.
The epidemiology of high dose ICS utilization
Despite ample guideline evidence emphasizing the utilization of low-moderate dose ICS, high-dose ICS prescribing remains common in real-world practice settings. Specifically, in patients with severe asthma, 24% of patients are on supratherapeutic doses (>1600 micrograms per day) prior to receiving biologic therapy.35
The dilemma: escalate ICS dose vs add-on therapy
Clinicians commonly face a practical dilemma when asthma remains uncontrolled on low-to-moderate dosage of ICS. There might be a pressure to escalate to high-dose ICS or to maintain the ICS and add-on adjunct therapy such as a long-acting muscarinic antagonist, a long-acting beta agonist, a leukotriene receptor antagonist or phenotype directed biologic therapy. Although dose escalation of ICS is straightforward, the incremental clinical benefit beyond moderate dosing is usually not worth the potential adverse effects associated with supratherapeutic dosages.
Conclusion
In summary, there is no evidence to support the use of high dose inhaled corticosteroids when compared to low-moderate dose inhaled corticosteroids in the current medical literature. There was no significant improvement in lung function, and there was no significant reduction in asthma exacerbations when the two dosage ranges were compared. Any potential benefits of high dose ICS are minimized by their adverse effects. To minimize the dose escalation of ICSs, the current literature recommends the use of additional agents such as LTRAs or LABAs. Hypothesis: Among patients with asthma who remain uncontrolled despite low-moderate doses of ICS with optimized adherence and technique, add on therapy with a secondary agent such as LABAs, LTRAs or LAMAs will be more effective at reducing exacerbations and controlling symptoms when compared to high-dose ICS, with fewer systemic adverse effects.

_total_daily_doses_for_adults_and_adolescents_by_device_formul.png)
_total_daily_doses_for_children_6--11_years_by_device_formulat.png)
_total_daily_doses_for_adults_and_adolescents_by_device_formul.png)
_total_daily_doses_for_children_6--11_years_by_device_formulat.png)