Introduction
Nearly 11 million Americans suffer from chronic wounds related to acute and chronic medical conditions.1 Chronic wounds may lead to serious complications, including infection, the need for recurrent surgical intervention, limb loss, and even death. Over the years, various biological wound matrices have been explored for the management of wounds, including fish skin. Decellularized fish skin when grafted to human tissue, acts as a scaffold for cellular infiltration, angiogenesis, and tissue regeneration.
KerecisTM, an Icelandic biotechnology company, has engineered wound healing products based on decellularized fish skin enriched with omega-3 fatty acids for use in cellular therapy and tissue restoration.2 These fish skin grafts are acellular matrices derived from the North Atlantic cod fish.2 Thanks to the anti-inflammatory properties of polyunsaturated fatty acids, fish skin xenografts have shown great promise in wound healing when compared to traditional wound management techniques such as autografts, allografts, and standard wound care involving frequent debridements and dressing changes.3 Fish skin xenografts are sustainable, accessible, and economically sound. Furthermore, fish is considered kosher and halal in respective cultures, making these grafts culturally acceptable to a wide range of patients.2 The following case presentation illustrates the clinical effectiveness and multifaceted advantages of fish skin xenografts in the management of complex wounds.
Case Presentation
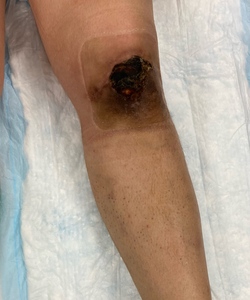
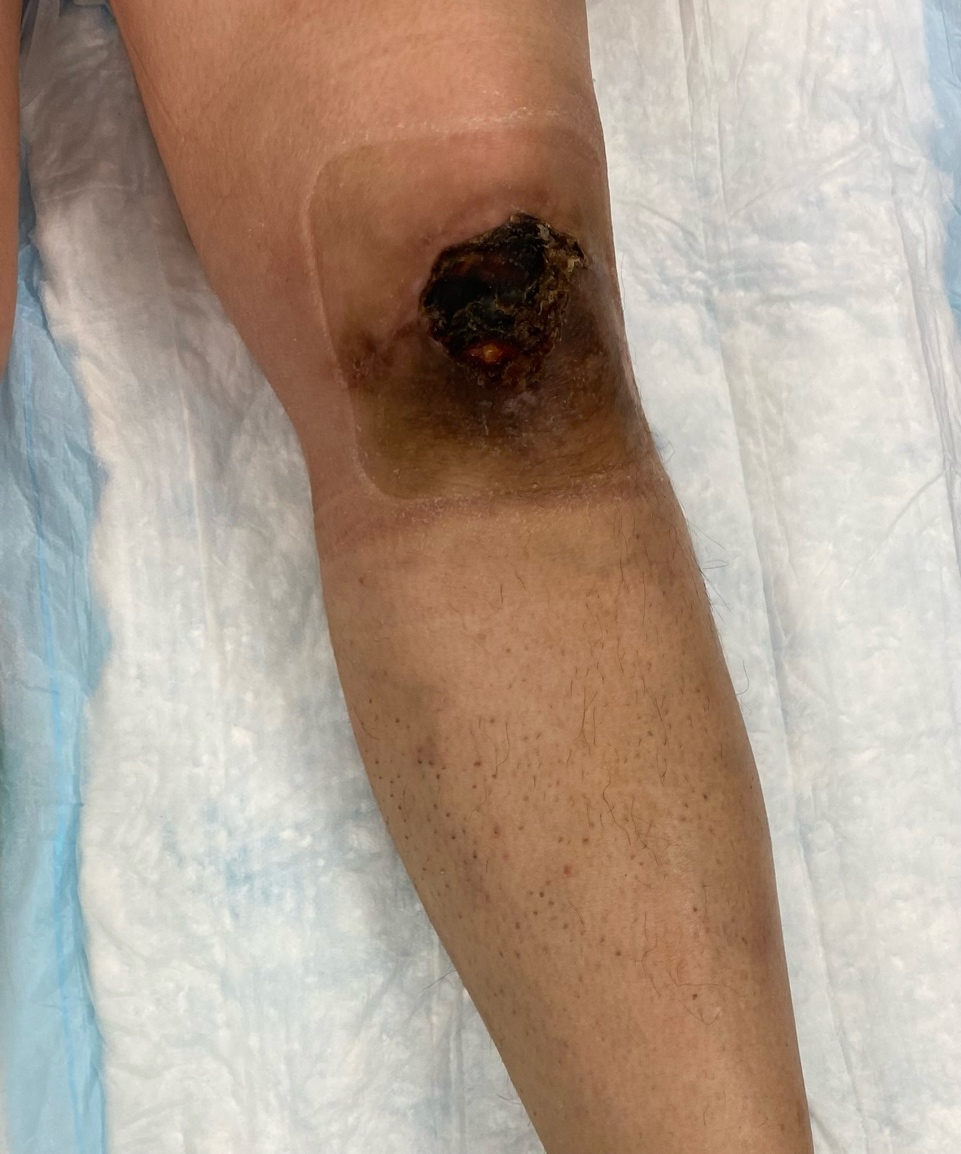
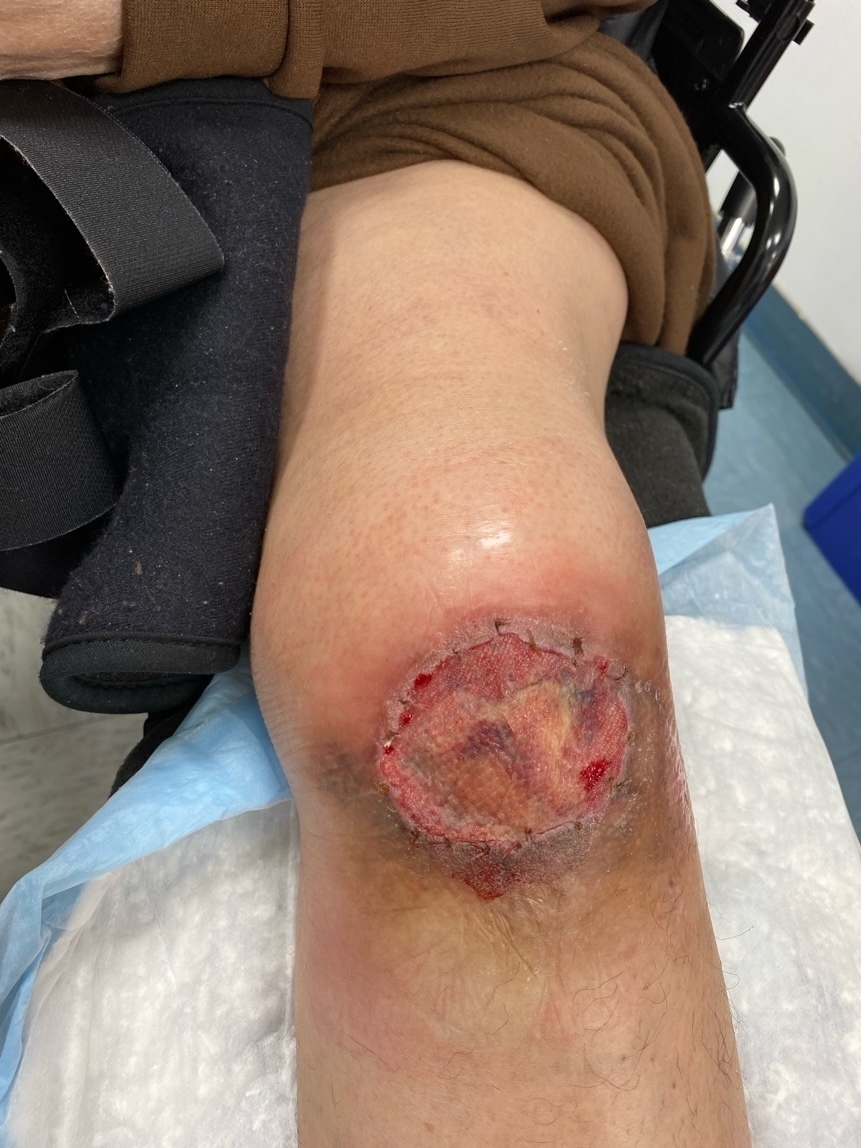
A 53-year-old female presented with a 6.5 x 5 x 0.1 cm non-healing left knee wound, with exposed patella, tendon, and hardware. The wound had persisted for approximately seven months following open reduction and internal fixation of a left patella fracture with patellar tendon repair. Her postoperative course was complicated by a surgical site infection requiring several months of intravenous and oral antibiotics, as well as serial sharp excisional debridements. Although the infection resolved, a chronic wound developed, with skin necrosis, ulceration, and eventual eschar formation (Figure 1).
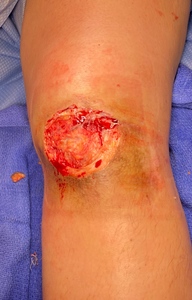
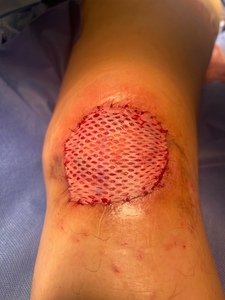
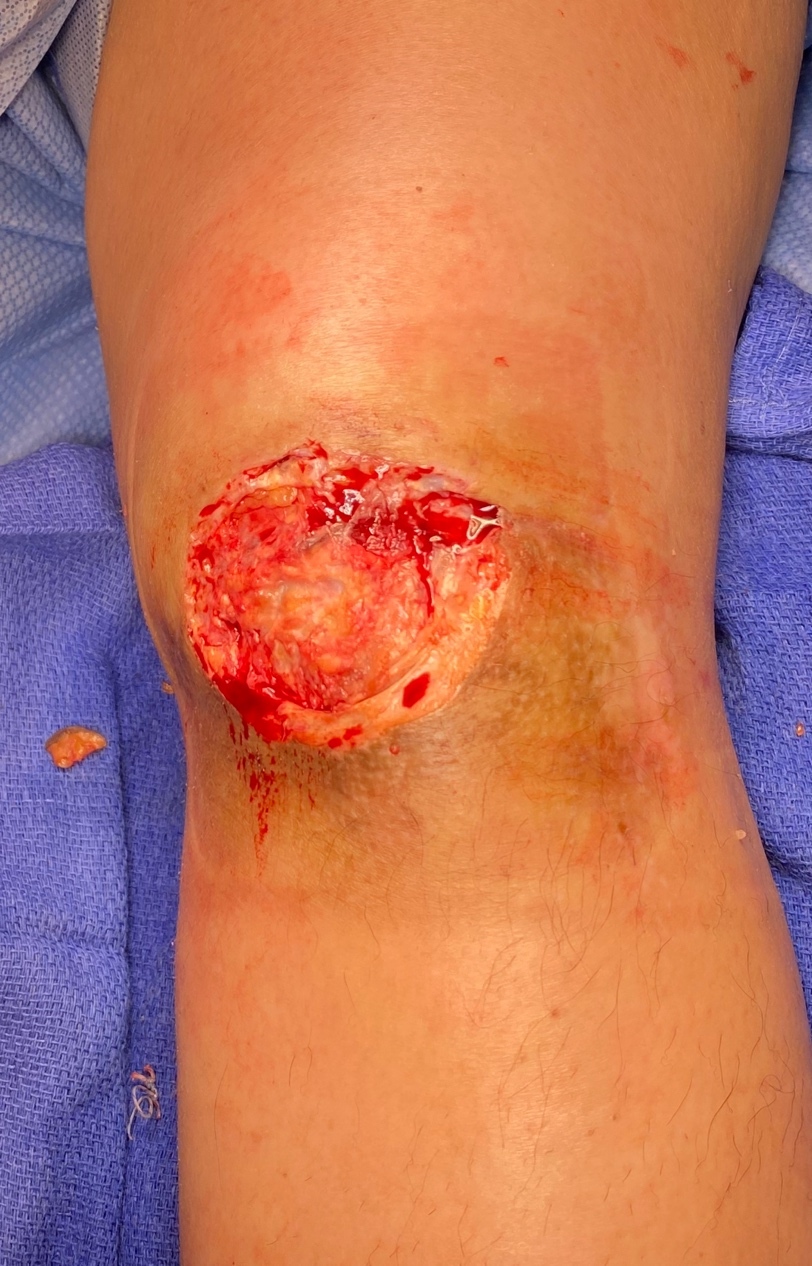
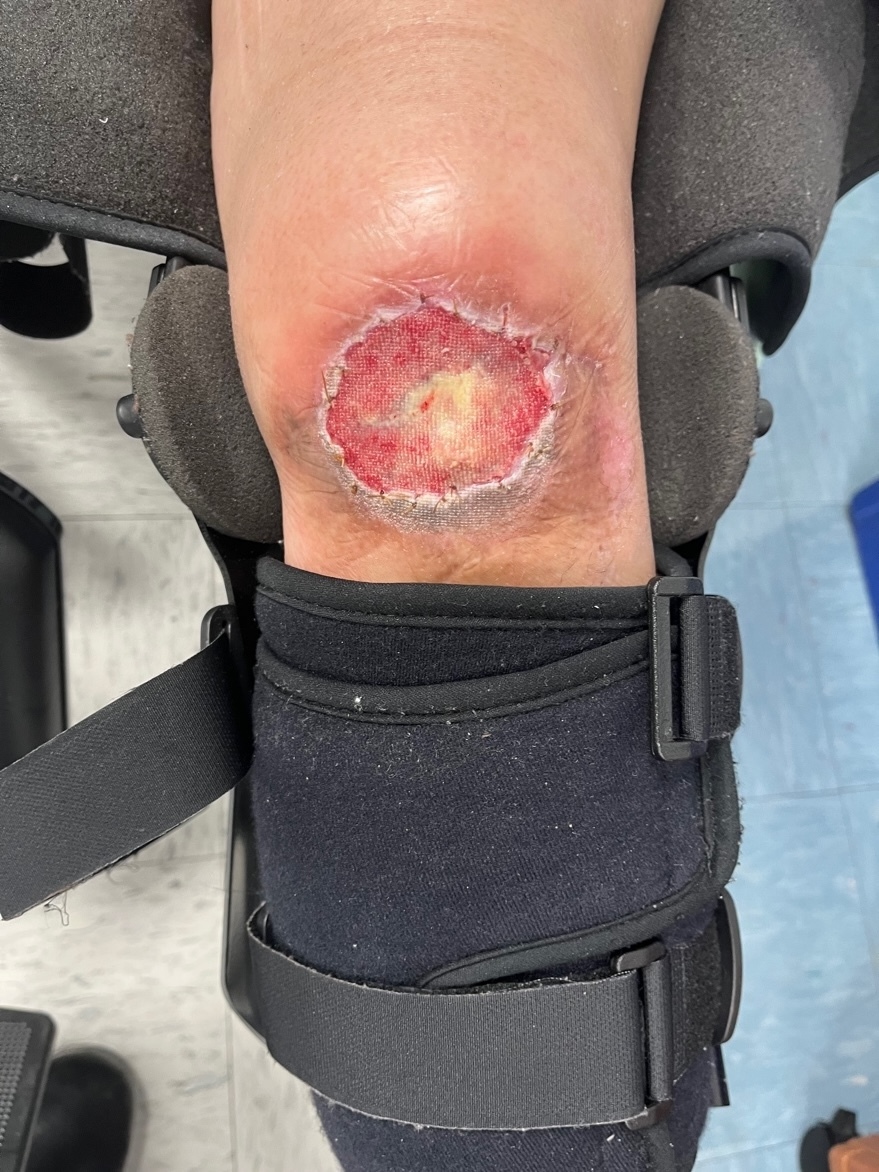
The patient underwent multiple excisional debridements over the course of four months until a decision was made to utilize Kerecis™ fish skin graft. Prior to application of the graft, the wound was tangentially debrided down to the central portion, followed by assessment of bleeding and tissue viability (Figure 2). Necrotic, exposed portions of the patella tendon were also debrided. To further improve vascularization of the Kerecis™ graft, the inferior edge of the wound was de-epithelialized.
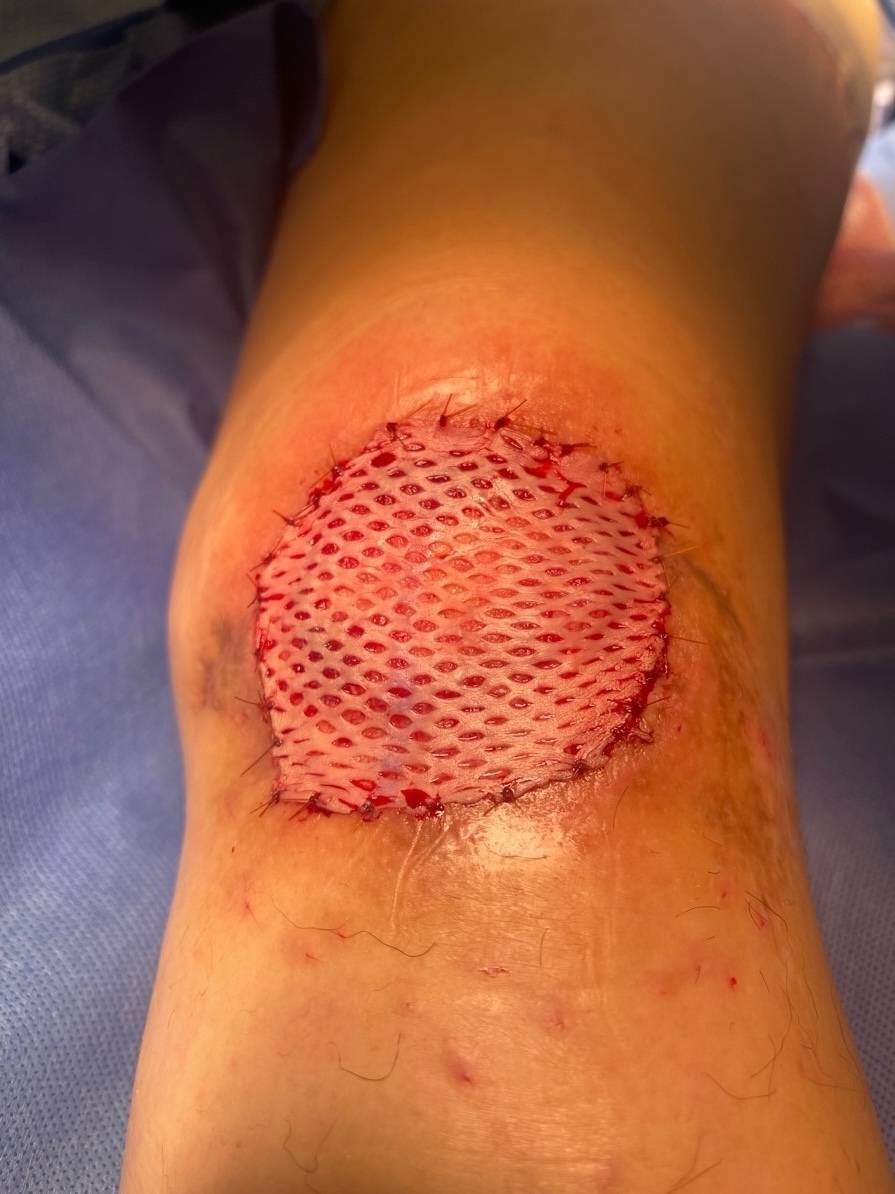
A Kerecis™ SurgiClose 7 x 10 cm solid graft was fenestrated with a scalpel and then hydrated using 20 cc of venous blood collected from the wound bed. The graft was affixed with chromic gut sutures and covered with 3MTM Adaptic Non-Adhering Dressing. A negative pressure wound therapy device was applied and set to 100 mmHg to enhance adherence of the graft. The patient was discharged home the same day with a knee immobilizer and minimal ambulation instructions to limit mechanical stress at the wound site and improve the probability of graft take.
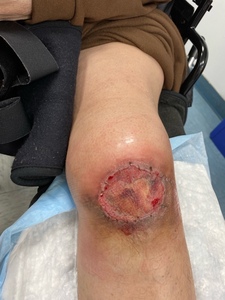
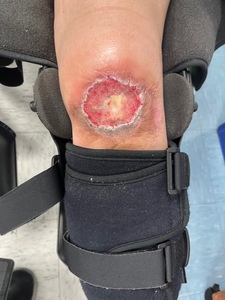
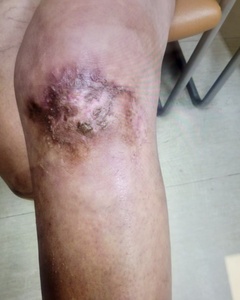
One week after graft application the wound bed showed improved vascularity (Figure 3). By two weeks, robust granulation tissue had formed (Figure 4). The patient had expected postoperative pain, which was controlled with over-the-counter acetaminophen and ibuprofen. There were no signs of infection throughout the recovery period.
Given the healthy, well-vascularized wound bed, the patient underwent definitive coverage with a split-thickness skin graft approximately nine days after her second postoperative visit. The graft was secured with chromic sutures and a vacuum-assisted closure (VAC) device set to 125 mmHg (Figure 5). Once again, the patient was discharged home the same day with a knee immobilizer and ambulation restrictions.
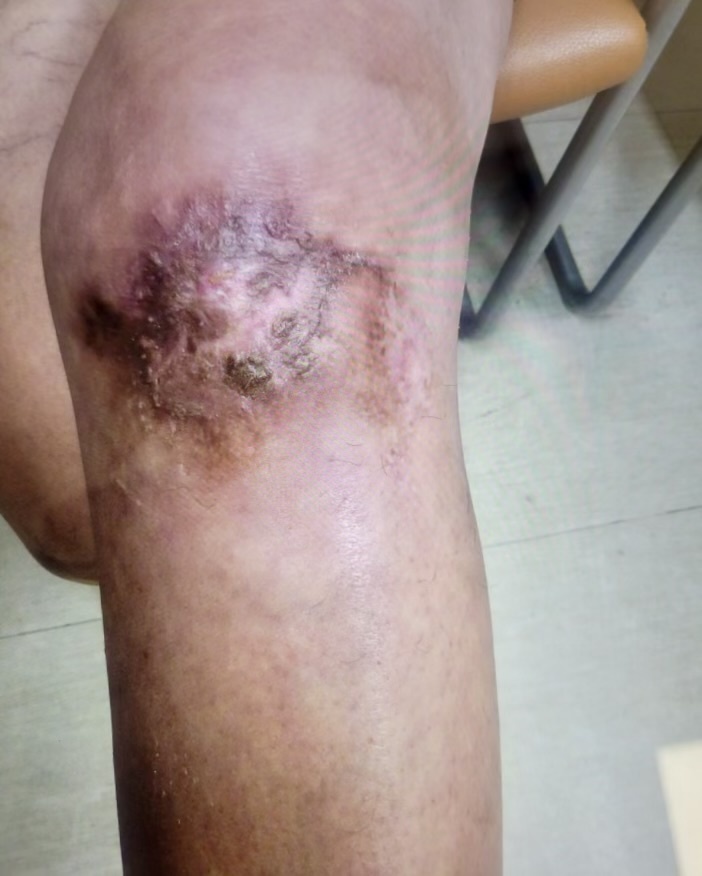
Postoperatively, the patient did well, without complications, pain was well-controlled, and there were no signs of infection. The skin graft demonstrated 100 % take, and the donor site was healing without complications. At two-month follow-up, the wound had fully healed, the patient was pain free, and she returned to full functional activities (Figure 6). Her outcome underscores the multifaceted benefits of fish skin grafts.
Discussion
Fish skin has emerged as a promising biologic material for wound management due to its rich content of omega-3 fatty acids, lipids, and protein. These components contribute to its anti-inflammatory properties, pliability, and ability to augment wound healing. Fish skin stimulates the recruitment and proliferation of the body’s cells for tissue regeneration. Kerecis™ fish skin xenografts are FDA-approved for treating acute and chronic surgical wounds.3,4 The product’s porous microarchitecture closely mimics human skin, making it an ideal scaffold for tissue remodeling.2
Other biological advantages of fish grafts include rapid cell mobilization and proliferation, inflammatory cytokine reduction, and natural bacterial resistance–largely attributed to the abundance of polyunsaturated fatty acids.3 These xenografts are biocompatible and have little to no infection risk due to their composition, enabling their approval for use in chronic wounds, burns, hernia repairs, and breast reconstruction.4 The application of the KerecisTM graft in this case presentation resulted in a substantial and reliable amount of granulation tissue formation within a short period of time, rendering it a valuable option in chronic wound management or possibly a bridge to more definitive reconstruction.
Despite their clinical advantages, cost remains a substantial drawback to fish skin xenografts. One square centimeter of Kerecis™ graft costs $154.91 per unit.3 This high unit cost can present a significant barrier for lower-income patients or those requiring repeated applications. However, one study conducted a cost analysis of using the Kerecis™ product against annual healthcare costs and found that annual healthcare costs decreased over time due to faster wound healing, thus reducing long-term care requirements.4 Another study reported an average wound healing cost of $3,927 per wound in the outpatient setting, while more complex wounds such as jeopardized flaps and grafts averaged $9,358 per wound.3
Although there is a financial burden with the use of fish skin grafts, their proven clinical efficacy and potential to lower long-term treatment costs support their continued integration into advanced wound care management.
Conclusion
The application of the Kerecis™ fish skin graft yielded remarkable results in this patient with a chronic, non-healing wound. This outcome highlights the anti-inflammatory and angiogenic properties of the product, as well as its efficacy in promoting healing in complex wounds. Moreover, successful application of a fish skin xenograft in this case is one possible broader potential in managing a wide variety of wounds, particularly chronic, complex wounds. Fish skin xenografts may perhaps serve not only as a definitive treatment, but also as a bridge to other management modalities such as skin grafting or flap reconstruction. As research in biologic scaffolds grow, further investigation is warranted to better define their indications, optimize patient selection, and evaluate long-term outcomes.
Disclosures
The author reports no financial disclosures or conflicts of interest related to this work.
Ethics Statement
Written informed consent was obtained from the patient for publication of this case along with accompanying images.