Background
Anti-GBM (anti–glomerular basement membrane antibodies) glomerulonephritis, also known as Goodpasture’s syndrome is a rare and severe autoimmune disease characterized by small-vessel vasculitis.1 Although its occurrence in patients with malignancy remains uncommon, anti-GBM disease is increasingly recognized in oncology practice with the widespread use of immune checkpoint inhibitors.2 It presents as rapidly progressive acute kidney injury, requiring early diagnosis.3 Anti-GBM disease in patients with malignancy remains rare,4–6 but some cases describe the disease being triggered by immune checkpoint inhibitors such as nivolumab or pembrolizumab, used in oncology.6 The case presented illustrates the complexity of diagnosis and management in this context.
Case presentation
This is the case of a 57-year-old patient with no history of diabetes, hypertension or tuberculosis, he was a chronic smoker since the age of 16, and quit five years ago. In 2018, he underwent a left lobectomy and thymectomy for pulmonary neoplasia. In July 2022, he was diagnosed with a local recurrence associated with cerebral, bone, and secondary adrenal metastasis, requiring Chemotherapy (Bevacizumab, Carboplatin, Paclitaxel, Docetaxel), radiotherapy, and immunotherapy (Atezolizumab, Pembrolizumab)
A year later, he was admitted to our department for the management of advanced kidney failure (KF), which was discovered incidentally during a follow-up evaluation for his illness.
On clinical examination, he was conscious, clinically stable, and afebrile (36.5°C), with no lower limb edema and preserved diuresis. Urine dipstick analysis revealed proteinuria (2+), hematuria (4+), and negative leukocytes and nitrites. However, he presented with diffuse arthralgia affecting both upper and lower limbs.
The biological workup showed a blood urea level of 1.54 g/L, blood creatinine level of 92 mg/L. , hyponatremia at 129 mmol/L, hypokalemia at 3.1 mmol/L, metabolic acidosis with bicarbonate levels at 17 mmol/L, hypoalbuminemia at 29 g/L, and 24-hour proteinuria at 2.97 g/day . Cytobacteriological urine examination showed leucocyturia at 63/mm³ and hematuria at 1128/mm³, but no germs.
The renal artery doppler ultrasound did not reveal any stenosis, and the kidneys were of normal size and well-differentiated.
However, there was a biological inflammatory syndrome with a C-reactive protein at 220 mg/L and procalcitonin at 337 mg/L. The viral serologies (hepatitis B, hepatitis C, and HIV) were negative, while PCR Cytomegalovirus (CMV) was positive with a viral load of 1414 (log 3.15). He was then treated with ciprofloxacin (ciprofloxacine), ceftriaxone (rocephine) , metronidazole (flagyl) for 15 days then imipeneme/cilastatine (bacqure) and teicoplanine (targocid) for 10 more days and ganciclovir (cymevene) 50mg three days per week.
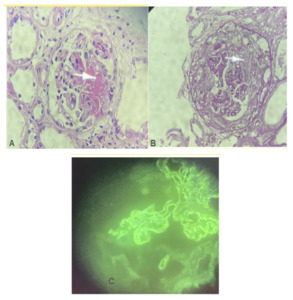
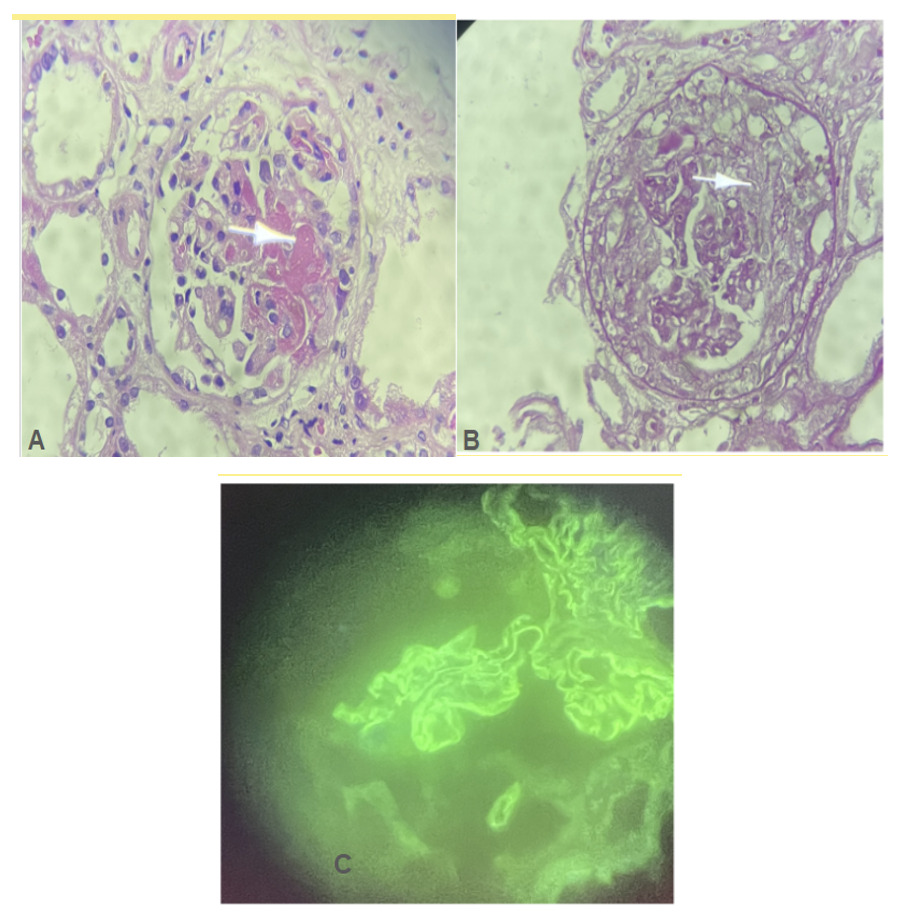
Immunologically, the anti-GBM antibodies were positive at 70 AU/mL, while anti-DNA antibodies, AAN, and ANCA were negative. The kidney biopsy confirmed diffuse necrotizing extracapillary glomerulonephritis with anti-glomerular basement membrane (anti-GBM) disease (LM: Fibrinoid necrosis, extracapillary proliferation in 5 out of 6 glomeruli with circumferential crescents, rupture of Bowman’s capsule, IF/TA < 5%; IF: diffuse linear glomerular deposits of IgG and IgA, segmental granular IgM deposits) (Figure 1).
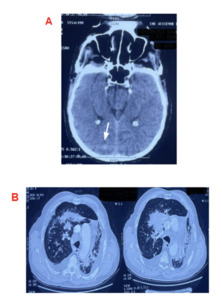
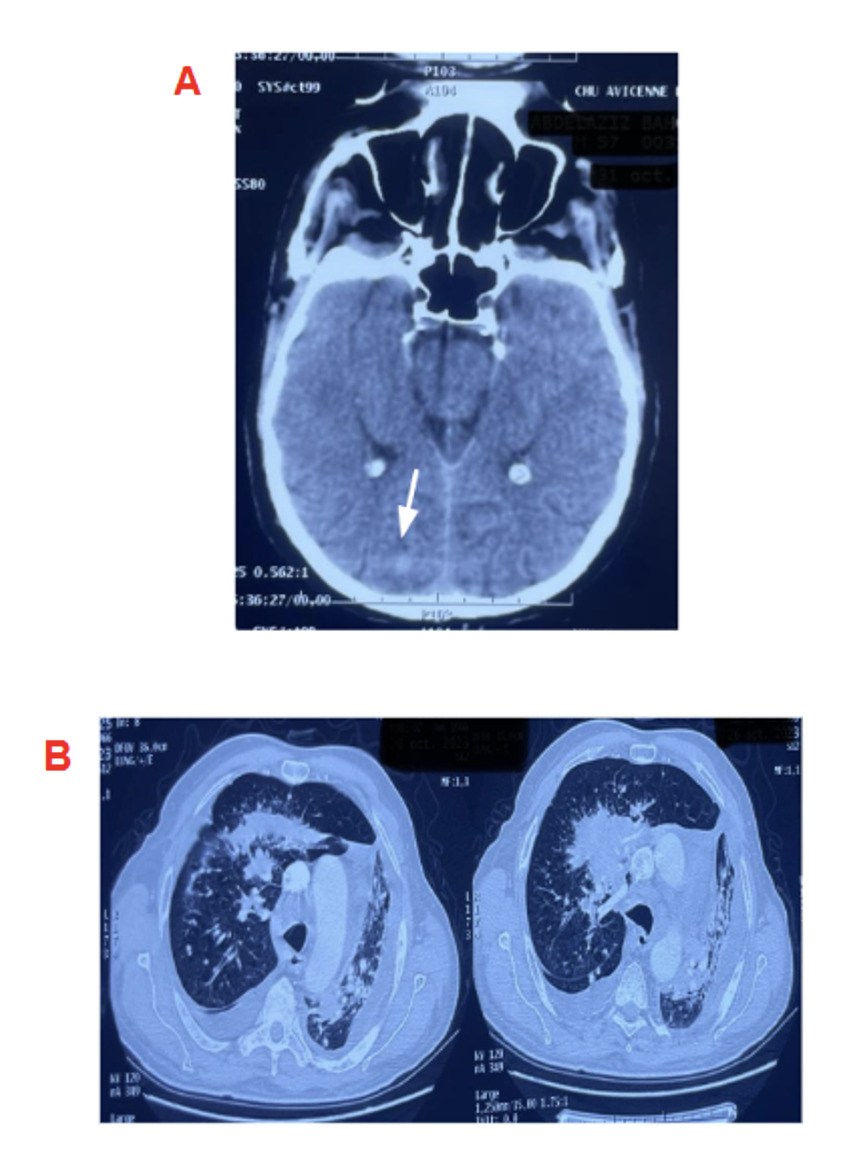
Regarding the etiology, the cerebral-thoracic-abdominopelvic CT scan revealed a cerebral mass, a pulmonary process with early carcinomatous lymphangitis, and secondary involvement of the left adrenal gland and bone metastases (Figure 2)
Therapeutically, he received 500 mg intravenous methylprednisolone (MP) boluses for 3 days, followed by oral corticosteroids (CS) (1 mg/kg/day) and hemodialysis was initiated via a tunneled right femoral catheter.
Four weeks later, the tumor showed progression with right basal alveolar hemorrhage, total collapse of the left lung field and moderate bilateral pleural effusion., with no current indication for surgical or pharmacological treatment. He was then transferred to intensive care for the management of a fulminant alveolar hemorrhage with intravenous methylprednisolone .
Despite the initiation of immunosuppressive therapy, including intravenous methylprednisolone pulses, oral corticosteroids, and hemodialysis, no improvement in renal function was observed, and the patient remained dependent on dialysis. The patient remained dialysis-dependent, but ultimately died from massive hemoptysis related to tumor progression, rather than directly from renal failure.
Discussion
Anti-glomerular basement membrane (anti-GBM) glomerulonephritis, also referred to as Goodpasture’s disease, is a rare autoimmune disorder characterized by the presence of circulating antibodies directed against the glomerular basement membrane. Clinically, it presents as a rapidly progressive glomerulonephritis and, in some cases, pulmonary hemorrhage.7 If not diagnosed and treated promptly, it can lead to irreversible renal damage and significant morbidity.5,8
Although rare, anti-GBM disease has increasingly been reported in association with malignancies and immune checkpoint inhibitor (ICI) therapy, suggesting a complex interplay between oncologic processes and autoimmune responses. Previous reports have highlighted cases where underlying malignancy, particularly renal cell carcinoma, may trigger anti-GBM disease, possibly by exposing cryptic antigens that elicit an aberrant immune reaction. For example, Rivedal et al. described an association between undiagnosed renal cell carcinoma and anti-GBM glomerulonephritis, suggesting a paraneoplastic mechanism.5 Similarly, El Yamani et al. reported the development of anti-GBM disease following administration of pembrolizumab, an ICI used in cancer immunotherapy.9 These findings underscore the necessity for heightened clinical suspicion in patients with malignancy or receiving immunomodulatory treatments who present with renal impairment.
The current case fits within this growing body of evidence, as the patient was undergoing chemotherapy and immunotherapy for metastatic malignancy involving the lungs, brain, and adrenal glands. The development of anti-GBM disease in this context likely reflects a multifactorial etiology, where both malignancy and immunotherapy may have contributed to a breakdown in immune tolerance. As previously reported, the onset of glomerulonephritis in such patients is often insidious but rapidly progressive, necessitating early recognition and aggressive management.1,3,10
In our case, the diagnosis was confirmed by the presence of circulating anti-GBM antibodies and characteristic findings on kidney biopsy, which showed diffuse necrotizing crescentic glomerulonephritis with linear IgG deposition. These histological features are consistent with classic anti-GBM disease and match descriptions from prior reports.11 Notably, mixed IgG and IgA deposits were also observed, raising the possibility of overlapping autoimmune phenomena, which may reflect the profound immune dysregulation seen in cancer patients receiving ICIs.3,9
Despite early initiation of high-dose corticosteroids and hemodialysis, renal function did not recover, and the patient remained dialysis-dependent. Standard treatment of anti-GBM disease consists of high-dose corticosteroids combined with plasmapheresis and additional immunosuppressive agents such as cyclophosphamide or rituximab. In our case, the use of aggressive immunosuppression was limited by active metastatic malignancy and concomitant CMV viremia. The clinical course was further complicated by the development of a fulminant alveolar hemorrhage, and the patient ultimately died from massive hemoptysis in the setting of tumor progression while remaining dialysis-dependent.12–15
Immunotherapy, particularly ICIs such as pembrolizumab, nivolumab, and tremelimumab, has revolutionized cancer treatment but is increasingly recognized as a trigger for immune-related adverse events, including anti-GBM glomerulonephritis.4,16,17 Between 2010 and 2023, multiple cases of ICI-associated anti-GBM disease have been reported, mostly in patients aged 50–75 years with solid tumors such as lung, cervical, or skin cancers (Table 1). Onset typically occurred within 2–4 months of initiating immunotherapy and was characterized by acute kidney injury, hematuria, and a systemic inflammatory response. Renal biopsies in these cases consistently demonstrated necrotizing crescentic glomerulonephritis with linear IgG deposition.
Management in these cases included withdrawal of the offending ICI, high-dose corticosteroids, plasmapheresis, and cyclophosphamide. Nevertheless, outcomes remained poor in the majority of reports, with many patients progressing to end-stage kidney disease or dying from pulmonary complications or cancer progression.
Our case shares many similarities with the reported literature but also presents notable distinctions. In particular, the detection of CMV viremia, the presence of IgA co-deposition, and the development of fatal alveolar hemorrhage point to a particularly aggressive and multifactorial immune response. These features underscore the need for a multidisciplinary approach to managing such complex cases, involving close collaboration between nephrologists, oncologists, and infectious disease specialists.
In conclusion, this case reinforces the emerging recognition of anti-GBM disease as a potential complication in cancer patients, especially those receiving immune checkpoint inhibitors. Early diagnosis, renal biopsy, and multidisciplinary management are essential. However, prognosis remains guarded, particularly in the presence of concurrent infections, advanced malignancy, or delayed treatment.
Conclusions
Anti-GBM glomerulonephritis is a rare condition, sometimes triggered by cancers or immune checkpoint inhibitors. Early diagnosis is crucial to prevent irreversible kidney damage. This case highlights the importance of better understanding the impact of immunotherapy on autoimmune kidney diseases and emphasizes the unique challenges faced by patients with metastatic cancers. Proper management requires a multidisciplinary approach to treat both the cancer and renal complications
Conflict of interest
The authors declare that they have no conflict of interest to disclose.
Ethics approval and consent to participate
The study was conducted in accordance with the ethical standards of our institution. The patient provided written informed consent for participation and publication. Ethics approval was waived by the local ethics committee due to the nature of the single case report and anonymized data.